2021上半年我国医疗器械出海步伐稳健
2021-11-18
新冠肺炎疫情爆发以来,对国际贸易和世界经济造成严重冲击,给全球公共卫生体系带来了挑战。我国生产的医疗防疫物资及相关医疗器械为全球抗疫贡献了重要力量,防疫用医疗器械出口快速增长成为我国对外贸易中的亮点。
根据中国医药保健品进出口商会(以下简称医保商会)统计,我国医疗器械2020年进出口贸易额突破1000亿美元,其中出口额约为730亿美元。

我国医疗器械出口额在疫情背景下持续大增,主要原因是在许多国家和地区医疗器械产业链、供应链受到重创甚至停摆之际,我国产业体系率先恢复正常运转,多年来沉淀的发展力量爆发,国际市场竞争优势进一步凸显。
2021年上半年,我国医疗器械产品出口与去年同期相比有所下降,主要原因是防疫物资中的口罩、防护服、呼吸机出口减少。但新冠病毒检测试剂出口增速较稳定,印度疫情促使制氧机出口激增,传统医疗器械产品出口恢复正常,医疗器械产品出口增速、出口产品结构逐步回归理性。
进出口市场保持健康发展 医保商会统计数据显示,今年1月 – 6月,我国医疗器械进出口贸易额为615.04亿美元,同比下降23.86%,其中,出口额443.88亿美元,同比下降31.32%;进口额171.16亿美元,同比增加5.96%。
出口额下降主要因为随着疫情防控常态化,全球市场对以口罩、防护服为代表的疫情防护耗材需求降低,出口额同比下降75.05%。同时,各个国家和地区疫情形势缓解,本土制造业有所恢复,对我国防疫产品的依赖程度降低。与疫情相关的影像诊疗设备出口同样略有下降,但其他传统及优势医疗器械出口恢复增长。

虽然出口额较去年明显减少,但纵观近几年我国医疗器械的进出口贸易发展趋势,医疗器械出口健康发展态势明显。值得一提的是,今年上半年我国医疗器械出口占比达53.53%,打破了以往西药类产品长期占据医药产品出口半壁江山的局面。
据统计,今年上半年,美国是我国最大的医疗器械出口市场,其次为德国、英国和日本,共占总出口额的49.09%。此外,“一带一路”沿线新兴市场也是我国医疗器械出口的重要区域,出口额为141.37亿美元,占比约31.8%,未来占比还将进一步提高。
从出口省份看,今年上半年我国医疗器械出口额排名前十的省份出口额占比高达89.92%,进一步体现出我国医疗器械出口的高度区域集中性,主要出口省份如广东、江苏和浙江均呈现明显增长态势,其中,广东出口额居首位。
从进口情况来看,今年上半年,我医疗器械进口额为171.16亿美元,同比增长5.96%。进口来源地仍以美国、德国、日本为主,占到进口总额的53.39%。进口产品中,导管类耗材、体外诊断试剂、高端医疗设备是主要品种。此外,核心零部件、关键原材料进口依赖度仍然较高。其中上海、北京、广州为医疗器械主要进口城市。

体外诊断及按摩保健产品表现亮眼
今年上半年,体外诊断产品成为出口增长最显著的医疗器械品种之一。为助力全球疫情防控,我国体外诊断企业积极开拓全球业务,出口额呈井喷式增长,与疫情相关的新冠病毒抗体、抗原、核酸等检测试剂受到全球市场青睐,检测试剂企业业绩整体向好。
今年上半年,在其他防疫物资出口走低的情况下,新冠病毒检测试剂产品出口依然增长显著。据医保商会统计,今年上半年,我国主要体外诊断试剂(HS编码38220010、30021500、38220090)出口额约为79.09亿美元,同比增长288%,福建、浙江、北京为我国体外诊断试剂出口主要省份。
其中,自测类试剂出口额为68.88亿美元,占比高达87.09%。主要出口市场集中在欧洲对新冠病毒自测类试剂产品有特殊授权的国家和地区,如德国、奥地利等。新冠病毒自测产品的大量国际市场需求,是推动新冠病毒检测试剂出口增长的主要因素。
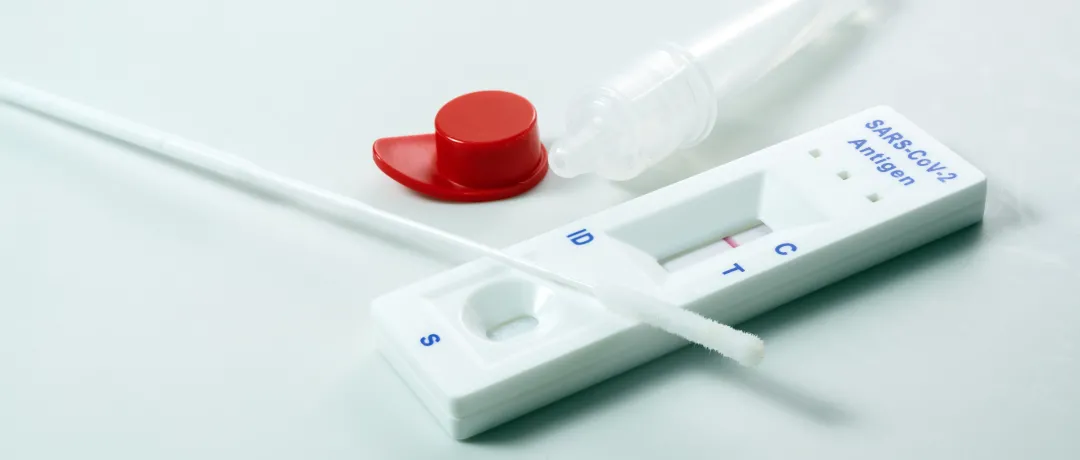
随着疫情防控常态化,很多企业的常规体外诊断试剂产品恢复出口,出口额也不断提升。目前,体外诊断试剂国际市场蛋糕被做大,尽快铺设海外营销渠道,并在出口地进行本土化推广已经成为我国企业开拓国际市场的重要任务。
受同质化严重、低价竞争激烈、国内集采价格压力影响,体外诊断行业“内卷”现象严重,布局出海对推动产业结构升级、提升行业竞争力和影响力具有重要意义。
根据Markets and Markets报告的数据,全球体外诊断市场规模预计将从2020年的845亿美元增长到2025年的960亿美元,年复合增长率为2.6%。

全球体外诊断市场分布并不均衡,发达经济体占据主要市场份额,如美国占有44.00%的市场份额,西欧占21.08%,日本占9.00%,合计占比高达74.08%。发达国家和地区市场已进入相对稳定的成熟阶段,其增长已呈现放缓态势。
而在发展中国家和部分地区,体外诊断作为新兴产业呈现出基数小、增速高等特点,预计在以印度、拉美等为代表的新兴市场中,体外诊断市场规模增速将会保持在15%~20%,新兴市场将会成为我国体外诊断产品企业的海外主战场。
而受疫情居家的影响,全球“宅经济”快速发展,家用医疗保健产品出口增长强劲。
2020年初受疫情影响,我国家用医疗保健产品出口一度受到重创,但依托产业多年的不断创新、持久沉淀和发展韧性,我国企业克服困难,顶住压力,实现了全年出口逆势上扬的佳绩。
今年上半年,按摩保健器具作为我国医疗保健产品中的传统优势出口产品,出口保持高速增长。根据医保商会统计,我国按摩保健器具今年上半年对外出口额高达29.60亿美元,同比增长86.77%,共出口200个国家和地区,尤其美国、德国、韩国、日本和英国对我国相关产品的需求呈现出明显增势,目前市场仍供不应求,预计今年出口额将达50亿美元。
国际化发展之路机遇与挑战并存
2021年,医疗器械继续占据我国医疗产品出口主导地位。国际市场对我国防疫物资的需求有所下降。
但与此同时,欧洲国家和地区对新冠病毒自测类试剂产品的特殊授权,使得国际市场对我国新冠病毒检测试剂产品的需求量相对平稳,疫苗预灌装注射器等相关产品需求量上升,印度疫情使印度对我国制氧机的需求激增,市场周期性波动显著,预计医疗器械出口额与2020年相比会略有下降,进出口总额在1000亿美元左右,欧盟和美国等传统出口市场,以及“一带一路”沿线新兴市场将是重要出口区域。
未来几年,我国医疗器械国际化发展将面临以下风险和挑战:一方面,由于国外经济增长速度普遍下滑且市场受防疫物资挤占,国际市场对普通医疗物资的采购能力有所下降。
另一方面,受疫情影响,工程师及销售人员出境受限,对深入海外开展市场推广活动及部分出口大型设备的售后服务工作产生了很大影响,不利于业务推广及销售活动。此外,防疫物资运输需求的激增还将导致集装箱流转速度下降,造成物流成本增高。原材料和人工成本上涨、跨境电商平台监管趋严等,也会进一步挤压企业出口利润。

从短期来看,在疫情影响下,全球对中国医疗器械供应链的依赖度提高。从长期来看,新冠肺炎疫情成为重塑全球医疗器械产业格局的催化剂。随着各个国家和地区鼓励本土产业发展政策的出台,国际竞争将越来越激烈,我国产品在全球市场中将面临更大的竞争挑战。但与此同时,我国对公共卫生健康问题的重视程度进一步加深,医疗大健康行业迎来发展黄金期,中国医疗大健康产业的国际地位会进一步提升。
来源:中国医药报










