医用敷料行业现状与研究方向分析,新型医用敷料将迎发展期
2021-06-18
一、医用敷料行业概况
狭义的医用敷料是指在伤口愈合过程中,可以替代受损的皮肤起到暂时性屏障作用,避免或控制伤口感染,起到保护创口、创面,提供有利于创面愈合环境的医用卫生材料,即伤口护理产品。广义的医用敷料则不仅包括伤口护理产品,还包括感染防护产品及消毒清洁产品等。
从发展历程来看,医用敷料从低附加值的传统医用敷料向高价值的新型医用敷料发展。
医用敷料行业发展历程
资料来源:华经产业研究院整理
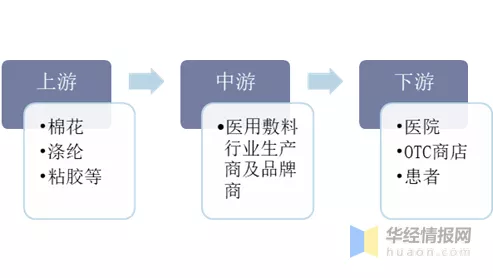
医用敷料行业的上游主要是棉花种植业和纺织行业;中游生产商、品牌商形成地域分化,产业链持续转移,代工生产商多集中于发展中国家,大型跨国品牌商多位于发达国家;下游是各级医院、疾控中心、OTC药店等,最后流通到患者手中。
医用敷料行业产业链
资料来源:公开资料整理
二、医用敷料行业现状
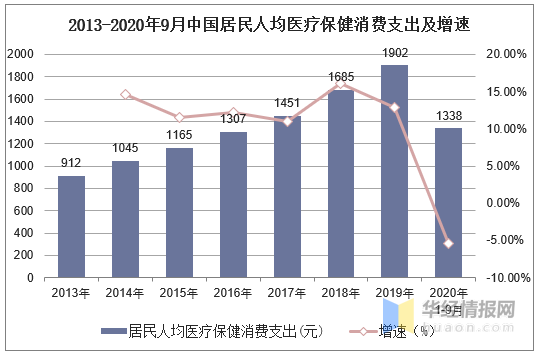
卫生意识强化、老龄化趋势下人均医疗支出、入院人数及手术次数提升,驱动医用敷料行业持续扩容。据国家统计局数据显示,2019年中国居民人均医疗保健消费支出达1902元,同比增长12.88%,2020年1-9月,人均消费支出为1338亿元,较2019年同期下滑5.37%。
资料来源:国家统计局,华经产业研究院整理
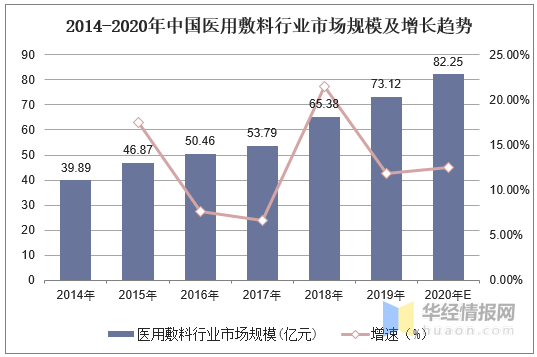
近年来,随着我国医疗条件的改善和居民健康意识、消费水平的提升,我国医用敷料市场稳步增长。2019年,我国医用敷料市场规模约为73.12亿元,预计到2020年,我国医用敷料市场规模将突破80亿,达82.25亿元左右。
资料来源:公开资料整理
2014-2019年,中国医用敷料出口规模呈先下降后上升趋势,2019年,中国医用敷料出口规模达27.2亿美元,占全球市场比重为21.8%。目前,中国已经成为全球医用敷料第一大出口国。
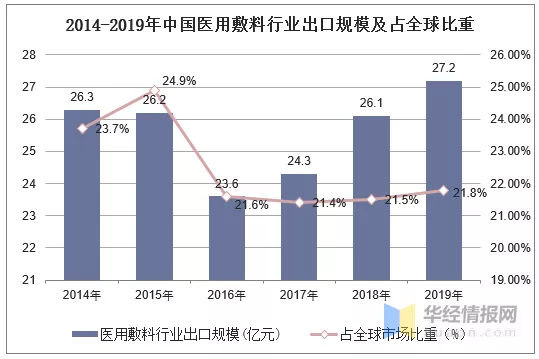
资料来源:中国医保商会,华经产业研究院整理
相关报告:华经产业研究院发布的《2020-2025年中国医用敷料行业发展趋势预测及投资规划研究报告》
三、医用敷料行业壁垒分析
从国内市场的竞争情况来看,由于我国医用敷料行业进入门槛较低,虽然市场容量较大,且增长较快,但行业内生产企业众多,且大部分为规模很小的地区性小企业,造成行业集中度较低。国产医用敷料以传统伤口护理类产品为主,产品同质化严重,国内企业多在低端市场内进行低价竞争,品牌识别度和影响力相对较低。

资料来源:公开资料整理
四、医用敷料行业研究方向
现阶段,世界上一些地区仍然频繁发生战争,人类还是面临着自然灾害、恐惧袭击的威胁,流血往往是司空见惯的事情,所以临床上研究止血敷料具有重大的现实意义。对于传统敷料而言,虽然具有保护与吸湿的功效,但在愈合时却很容易造成伤口粘连,进而使二次损伤发生,加之还有诸多不足存在,如缺乏良好的透气性能,不能很好的止血等。经过发展,一些新型敷料,如无纺布、止血粉等在多个方面取得了显著进步,包括透气性能上、免伤口黏连、止血时间上。今后敷料的发展重点将集中在:积极研究止血敷料;止痛、促进创伤修复方面;在储存上,要求可与海水、低温、高温等各种恶劣环境相适应;研究中药辅料(三七、白芨、云南白药);另外,智能敷料也不断出现,日本率先推出了富含离子型活性药物,利用渗出液对活性药物予以有效抑制,加快药物药效发挥。经济社会高速发展,社会大众十分注重自身健康,对敷料的要求更加严格,对于此,我们应积极研究战时急救敷料与创伤救治敷料,以充分保障人类的生命安全。
来源:华经产业研究院











