突围战:国产内镜将走向何方?
2022-01-19
2015年9月,国家层面发布《关于推进分级诊疗制度建设的指导意见》作为建立分级诊疗制度的总纲领,明确提出“引导优质医疗资源下沉,形成科学合理就医秩序,逐步建立符合国情的分级诊疗制度,切实促进基本医疗卫生服务的公平可及。”
推行至今,基本建立“基层首诊、双向转诊、急慢分治、上下联动”的分级诊疗制度,这个目标实现了吗?
数据显示,2012至2018年间基层医疗机构诊疗人次绝对值逐年上升,但相反基层医疗机构占比逐年下降。“分级诊疗”效果并不尽如人意,大医院人满为患、基层医院“门可罗雀”的状况仍未根本缓解。
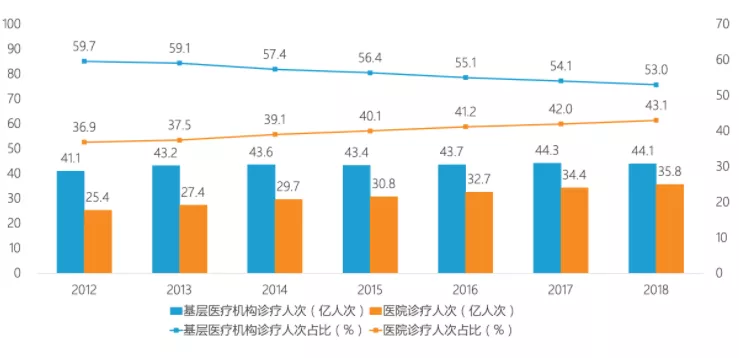
数据来源:国家统计局,图源:亿欧网
如何推进分级诊疗落地,问题的症结究竟在哪里?
01
推进分级诊疗制度,需重塑基层医疗
政策脉络:「郡县治,天下安」。在中国,县域人口占全国人口70%以上,县级基层医院占全国医院总量近半数,是我国医疗服务体系中承上启下的关键枢纽,承担着健康守门人的重要角色。
然而,由于优质医疗资源短缺与分布不均,基层医疗也是我国医疗体系的薄弱环节。设备配置不足、专业人才短缺、专科能力建设缓慢……成为长期制约基层医疗机构发展的痛点,提升基层医疗水平迫在眉睫,“分级诊疗”一词被推至幕前。

根据公开资料查询,图源:亿欧网
02
以“强基层”为重点工作
要构建起强大的公共卫生体系,重中之重,是需要强化基层。
《关于推进分级诊疗制度建设的指导意见》提出了以“强基层”为重点完善分级诊疗服务体系,明确基层建设是构建分级诊疗的基础,并提出六大保障措施,通过给予人才激励保障政策、提升县医院综合能力等方式对基层的倾斜支付政策,转变患者“凡病就到大医院“的思想,落实“基层首诊”。
这意味着,基层医疗机构的功能主要是向基层人民提供基本公共卫生服务和基本医疗服务,为诊断明确、病情稳定的慢病患者提供服务,“将常见病留在基层医疗机构诊疗,将疑难杂症转诊到上级医院进一步进行治疗”。
在政策的“保驾护航”之下,基层医疗机构的数量近年来增速加快,2019年已达96万个;基层医疗机构床位数得到保障,2012-2018年间呈现增长趋势;同时,万元以上医疗设备快速补充。
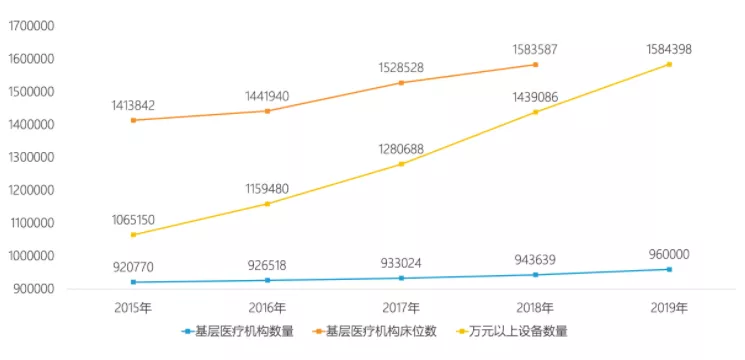
数据来源:国家统计局,图源:亿欧网
同时,“强基层”的概念提出后,全国多地纷纷响应。例如2021年9月,江苏省政府办公厅发布了《江苏省“十四五”卫生健康发展规划》,其中特别强调:到2025年,80%的乡镇卫生院和社区卫生服务中心达到国家服务能力基本标准,30%的乡镇卫生院和社区卫生服务中心达到国家服务能力推荐标准。
03
内镜遇上基层医疗:下沉,拓维,潜入深海
2021年,“十四五”全新开端,中国医疗卫生事业迎来加倍提速的历史拐点,构建分级诊疗新格局,推动优质医疗资源扩容和区域均衡布局,已成未来五年医疗卫生体制改革的核心命题,基层医疗被赋予全新使命。
我国属于消化道疾病高发地,消化疾病的控制和早发现主要依赖早期筛查,以日本为例,早期胃癌的内镜检出率可达70%,而我国比例仅为15-20%左右,90%以上的胃癌在发现时已是中晚期,给家庭和社会带来沉重负担。

图源:国元证券研报
长海医院消化内科主任李兆申教授曾表示:“这种现状很大程度上由基层检查手段不足、百姓防治意识不够造成。”并提出“防治胃癌应重心下移,沉入基层。”临床上最为常见的诊断性胃肠镜难度较低且培养医生周期较短,完全可以在基层医疗机构完成,然而,基层地区因受医疗条件所限、专业诊断医师不足,大批的患者仍然涌向三甲医院进行胃镜或肠镜检查,倍受疾病和奔波之苦。
在分级诊疗的大背景之下,提高内镜诊疗在基层医疗机构的渗透率,合理配置医疗资源提高利用效率将是我国现阶段的目标,此举带来多层含义:
于基层医疗机构而言,将有机会获得更多内镜诊疗资源,承接基础内镜诊疗职能,内镜的渗透率和配置率加速提升;
于内镜企业而言,基层医疗机构将成为内镜设备的主要增量市场。据光大证券研究报告测算,2024年中国开展内镜诊疗医疗机构中,一级及以下的占比将提升至43%。广阔市场面前,如何以合适的产品去契合基层医疗机构的需求,值得各大内镜企业去深思与规划。
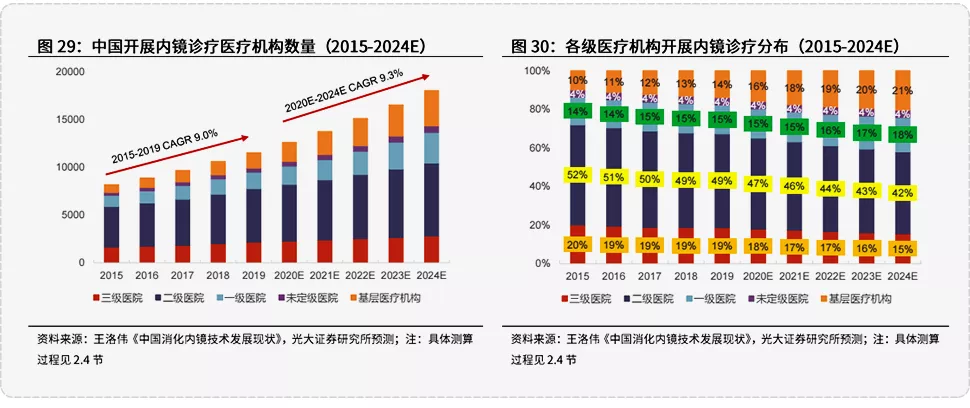
图源:光大证券研报
04
普惠基层,或成国产内镜破局之策
医用内窥镜按镜体是否可弯曲,分为硬管内窥镜和软性内窥镜两大类。由于行业技术壁垒高,目前,我国软性内镜市场被入局较早的三家日系企业——奥林巴斯、富士和宾得占据,总份额达90%以上。随着企业科技创新能力的提升与“国产替代”相关政策的出台,国内不少企业逆风扬帆、参与其中,如上海澳华内镜,早在2005年就推出了电子内镜,打开了该领域进口垄断格局的缺口,但由于进口日系企业体量相对较大,国产内镜市场仍有待突破。
“健康中国”下的普惠医疗。
“健康中国”作为国家的一项重大部署决策,早已深入人心,随着行动的深入实施,“普惠医疗”这一概念应运而生;目前,普惠医疗并没有官方定义,可直观理解为基本服务人人享有、人人保障,看得起病、看得好病。这与健康中国的理念是契合的,既是健康中国的内在要求,也是健康中国顺利实施、实现目标的必要途径和重要支撑。

图源:图源:《“健康中国2030”规划纲要》
普惠之风起,内镜诊疗却遭遇了“难题”。
让基层“痛苦”的话题——成本。
公开数据显示,日系企业主流的内镜系统价格超过百万元,据奥林巴斯年报显示,奥林巴斯的客户主要是三级医院和一小部分二级医院,基层医院的占比较低。
长期以来奥林巴斯的战略重心持续瞄准高端市场,对于国产企业来说,中国制造有基础,且有主场政策、成本等优势,有更大可能拿下基层医院广阔的内镜增量市场。
填补基层市场需求,成解决内镜“普惠化”关键。
成本是基层医疗机构无法回避的话题,如何将资金、人力、时间等多种成本的管理做到切实可控?动辄售价百万元以上的进口内镜设备匹配的“强大功能”是否真正契合基层医疗机构的基础需求?
有时“All in”并不意味着合适,也许,市场急需一款适配基层医疗的“普惠型”内镜来匹配不同层级的医疗需求,这将赋能基层医院,提升医院常见病、多发疾病和重大疾病诊疗能力,推动基层健康卫生事业新发展,做到真正意义上的资源下沉。在这个维度上,依托了创新资源、创新技术、创新手段、本土管理、高性价比等优势的国产内镜企业会更受青睐,并有望打破价格壁垒,将进一步加快国产替代的发展步伐。
内镜突围,非一朝一夕的事。
随着技术的发展以及内镜诊疗渗透率的提升,内镜市场将进一步提升,未来3-5年内,国内企业孰能在市场竞争中成功突围?
来源: 器械之家











