我国生物医用材料产业现状、机遇和新模式
2021-08-19
生物医用材料是一类用于诊断、治疗、修复或替换人体组织、器官或增进其功能的高技术材料,也称生物材料或生物医学材料。按国际惯例,其管理属医疗器械范畴。作为新兴产业,随着人口老龄化进程的加快、高新技术的持续注入,以及全球政府机构在政策及资金上的不断支持,生物医用材料产业得到了快速发展,并正在成长为世界经济的一个支柱性产业。我国在生物医用材料的研发、生产、应用上与国外先进国家有较大差距,且高端产品依赖进口,近年来一些优秀的生物医用材料企业陆续被国外巨头收购,产业层面的发展力量被进一步削弱。为了改善整体落后的局面,我国已出台多项政策支持生物医用材料的研发和产业化。生物医用材料产业正在快速追赶国际先进水平。
我国和全球生物医用材料产业发展比较
我国医疗器械行业规模增速远超全球平均增速,未来增长空间广阔。随着老龄化的加速、经济发展和生活质量的提高,以及人们对健康要求的日益提升,包括我国在内的全球医疗器械市场持续增长。Evaluate MedTech的报告显示,2012—2018年,全球医疗器械销售规模从3570亿美元增加到4278亿美元,年均增长3.1%(图1);预计到2024年将达到5945亿美元,年均增长约5.6%。前瞻产业研究院数据显示,2012—2018年,我国医疗器械销售规模从2687亿元增加到6071亿元,年均增长14.6%,是全球平均增速的近5倍。详见图1所示。根据中国医疗器械行业协会的统计,未来5年我国医疗器械市场复合增长率约为15%~20%,远超全球平均增速。中国新闻网的报道显示,截至2018,我国药械比(人均药品消费额/人均医疗器械消费额)仅为1:0.19,远低于全球1:0.7的平均水平,更低于发达国家1:1的水平。
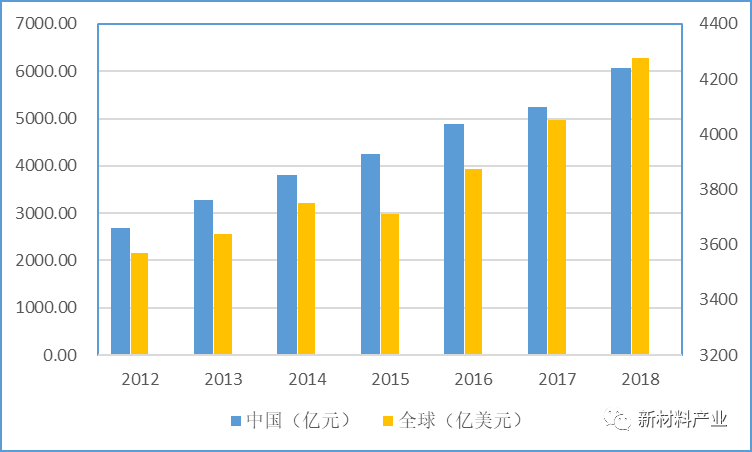
图1 2012—2018年全球和我国医疗器械行业规模
我国生物医用材料市场增速是全球的近4倍。全球医疗器械市场主要包括体外诊断、心血管、诊断影像、骨科、眼科、整形外科、内视镜、药物载体、牙科、糖尿病护理、伤口护理、健康信息系统、神经系统、肾脏系统、耳鼻喉科及其他等16个细分领域。其中,生物医用材料主要用于心血管类、骨科类、眼科类、整形外科类、牙科类、伤口护理类和肾脏系统类等7个细分领域的医疗器械产品。Markets and Markets数据显示,2016—2020年,全球生物医用材料市场规模将从1709亿美元增加到3000亿美元,年均复合增长率达到6%,远超同期全球医疗器械市场增速。全球生物医用材料约占医疗器械产品市场份额的40%~50%。2010—2016年,我国生物医用材料市场规模从670亿元增加到1730亿元,年均增长17%,预计2020年将达到4000亿左右,近5年年均增速达到23%,是全球生物医用材料市场增速的近4倍。
全球生物材料市场高度垄断,我国在部分领域实现了国产化。全球90%以上的医疗器械公司为中小企业,主要开展新产品、新技术研发,通过向大企业转让技术或被大企业兼并得到发展。大企业主要从事产业化及市场开发。2017年全球排名前20位的跨国公司占全球医疗器械市场的45%,尚无一家国内企业入围。心血管材料领域,排名前10的公司占有80%的市场,其中Medtrocnic、Abbott、Boston Scientific、Edwards LifeScience、Johnson&Johnson、Terumo等排名前6位的公司占有70%的全球市场;我国乐普医疗排名第9位,2017年全球市场占比约为1.4%。骨科领域,排名前10的公司占有88%的全球市场,其中Johnson&Johnson、Zimmer Biomet 、Stryke、Medtronic、Arthrex和Smith&Nephew等排名前6位的公司占有80%的市场,没有一家国内企业进入前10,但创生医疗器械(中国)有限公司、康辉医疗器械有限公司、威高集团有限公司、苏州欣荣博尔特医疗器械有限公司等已占有骨创伤修复器材国内市场的65%。Straumann、Dentsply、NobelBiocare和Osstem基本垄断了牙种植体和牙科材料。总体上,我国70%的高端生物医用材料仍依赖进口。
我国生物医用材料产业发展的机遇
旺盛的需求
随着全球人口自然增长、人口老龄化程度提高,发展中国家经济增长以及不断增加的医疗费用支出、日益提升的消费能力和健康意识,长期来看全球范围内医疗器械和生物医用材料市场将持续增长。世界卫生组织数据显示,2000—2050年,全世界60岁以上人口的数量将从6.05亿增长到20亿,占全球人口的比例将从11%增长至22%,欧盟、加拿大、中国60岁以上人口占比将超过30%。欧美日等发达国家和地区生物医用材料产业发展早,对产品的技术、质量要求高,需求主要是产品的升级换代,致使市场规模大,但增长较稳定。我国、东南亚等新兴市场是全球最具潜力的市场,需求主要是产品普及与升级换代,近年来增速较快。改革开放至今,我国已建成了门类齐全、独立完整的产业体系,成为世界制造大国。同时,在与国际同等技术条件下,“中国制造”成本低,性价比高,市场竞争空间巨大,我国具备了生物医用材料产业加速发展所需的市场空间和工业基础。
完善的政策
围绕创新研发、产业化、推广应用、贸易便利化、监管等方面,国家出台了一系列相关规划、政策,加快生物材料产业发展,详见表1。从顶层设计上,出台了《“十三五”国家战略性新兴产业发展规划》《新材料产业指南》以及配套的战略性新兴产业重点产品和服务指导目录(2016版),明确了发展方向和重点产品品种。为加快研发创新,科技部在国家重点研发计划中设立了“生物医用材料研发与组织器官修复替代”重点专项,2018年立项22个项目,支持经费达到2.3亿元,先健科技公司、冠昊生物科技股份有限公司等多家企业承担了相关任务。在产业化和推广应用方面,工业和信息化部批准建设“生物医用材料应用示范平台”,并采用保险形式支持重点新材料首批次应用。在贸易便利化方面,国务院批复“创新进出境生物材料监管”模式,广州生物材料通关周期由30天缩短到5天,上海以风险评估方式代替前置审批,评估时间缩短1/3。在监管方面,持续加大对医疗器械创新的扶持力度,加快审批速度、加快注册上市流程。
我国生物医用材料产业发展新模式
生物医用材料涉及学科交叉最多、资金和知识高度密集,需要生物材料企业、医疗器械企业、临床医院等上、下游的支撑,产业主要集中在经济、技术、人才发达或临床资源丰富的地区,行业集中度和垄断化程度不断提高,生产和销售日趋国际化。医工研企协同创新成为我国生物医用材料产业发展的新模式。
行业垄断化。为提升企业的市场竞争力,多数大公司已不再局限于最初的单一产品,而是通过企业内部自主创新和兼并收购,不断进行新产品的研发和产业化。而中小型企业则依靠向大公司转让新技术、新产品或被大公司兼并来维持生存,由此便形成了国外生物医用材料产业的“寡头”统治局面。
生产和销售国际化。国外几乎所有的生物医用材料企业均是大型跨国公司,其营业收入有相当多一部分来自国际市场。跨国公司通过向海外输出技术与资金、建立研发中心和生产基地,就地开展研发和生产,从而不断提升其在国际市场上的份额。
产业集聚化。生物医用材料发展对技术和人才素质要求高。生物医用材料产业多集聚在经济技术发达、人才富集或临床资源丰富的地区。例如,美国主要集聚于技术资源丰富的硅谷、128号公路科技园、北卡罗来纳研究三角园,以及临床资源丰富的明尼阿波利斯及克利夫兰医学中心等;德国主要集聚于巴州艾尔格兰、图林根州等地区;日本主要聚集于筑波、神奈川、九州科技园。我国主要集中在长三角、珠三角和京津环渤海地区。
医工研企协同新模式。生物医用材料产业具有高投入、高风险、技术领域广、研发周期长、流程环节多的特点,一般机构很难同时具备研发、临床、注册、生产、营销和售后所需的全部技术和能力。为了降低成本和风险,位于产业链不同环节、具备不同专业能力的企业和机构需要充分发挥专长,分工协作,优势互补,合作完成整个产业化过程。通过医学与工程技术的结合、研究机构与企业的结合是实现生物医用材料产业创新发展的新模式。
来源:赛迪智库材料工业研究所











