人工血管3——大盘点:上市人工血管汇总,明星产品特点分析
2022-01-28
导读
和义广业【行业分析】之人工血管系列,将深入分析人工血管的制备、临床应用、市场容量、国内外代表产品以及创新产品研究现状。本篇盘点汇总了中国和美国已上市的人工血管产品,并就代表性人工血管产品进行简介分析。
4.中美已上市人工血管产品
于NMPA进口医疗器械数据库中检索“人造血管”和“人工血管”(检索日期:2021年10月22日 ),数据如下:
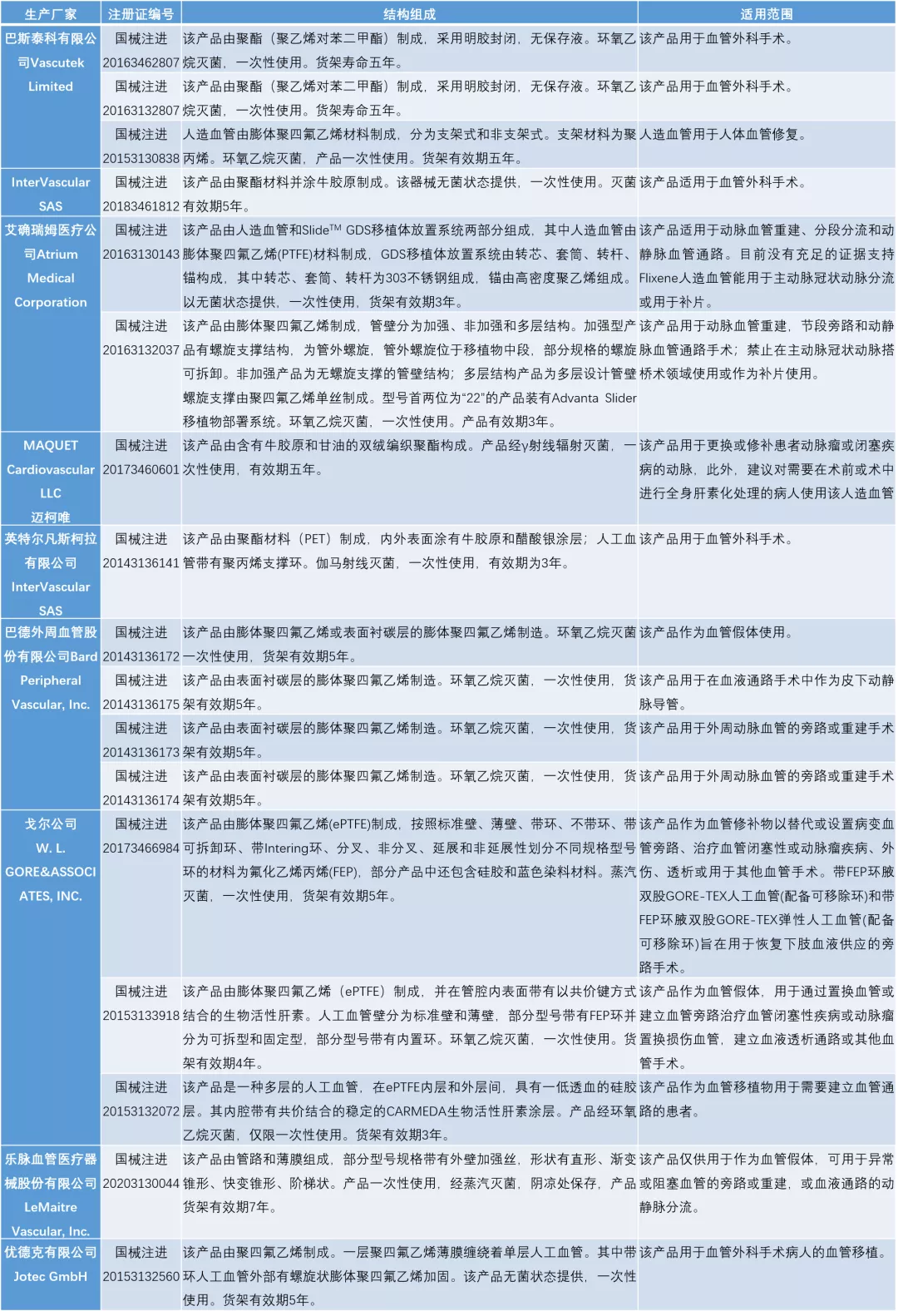
于NMPA国产医疗器械数据库中检索“人造血管”和“人工血管”(检索日期:2021年10月22日 ),数据如下:

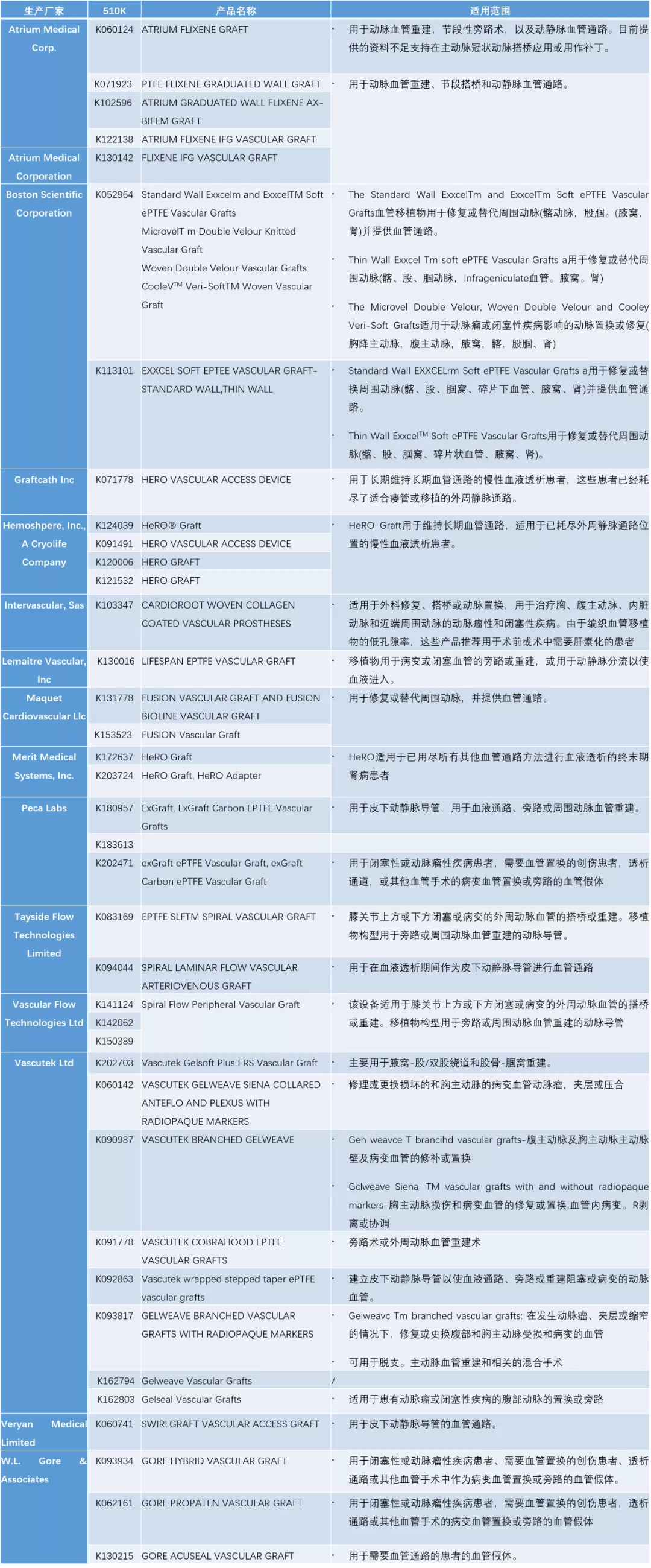
于FDA 510k数据库中检索“Vascular Graft”(检索日期:2021年10月22日),2006年-2021年登记数据如下:
Device Classification Name:
Prosthesis, Vascular Graft, Of 6mm And Greater Diameter
Prosthesis, Vascular Graft, Of Less Then 6mm Diameter(检索结果为零)
表. 2006-2021年 于“Prosthesis, Vascular Graft, Of 6mm And Greater Diameter”类别 取得510k的产品列表
5.上市人工血管重要品牌产品简介
1)MAQUET[1]
始于1838年德国,瑞典GETINGE AB集团旗下,拥有外科系统部/重症系统部/心血管外科部/麻醉系统部四个专业部门。
其人工血管业务是在收购中发展起来的,一次是在2008年收购波士顿科学的心血管外科业务,主要负责美国市场销售,2009年收购Datascope集团,负责在欧洲市场销售。后关闭美国工厂将生产基地搬到法国。

图. 产品:FUSION Vascular Grafts
产品构成:FUSION 血管移植物是由两层构成。内层由膨胀聚四氟乙烯 (ePTFE) 组成。肝素涂层通过白蛋白(人类血液中的主要血液蛋白质)粘合到该内层。外层由针织涤纶织物组成。这两层用专有的聚碳酸酯 – 聚氨酯粘合剂融在一起。
临床研究:于ClinicalTrials.gov检索“MAQUET”,结合公开文献,总结临床研究结果如下:


通过上述实验MARUET产品的涂层技术相比较单一聚四氟乙烯产品具有较好的长期血管通畅率。
2)Terumo
泰尔茂株式会社成立于 1921年,总部位于日本东京。其产品包括一次性医用器械、血管造影与治疗用导管、医用电子产品系列、人工心肺产品系列等。2002年10月收购英国制造和销售人工血管产品公司“Vascutek Co.”。
泰尔茂发明了独特的水解蛋白明胶预凝,无孔设计,斜纹编织,多分叉血管提高手术安全性,减少手术时间,聚酯血管和聚四氟乙烯血管。提供了胸腹部及四肢血管的多方面选择。
来源:和义广业创新平台











