EKF Diagnostics公司在1990年由Berthold Walter创建,起初只是一家技术性公司,发展缓慢,90年代后期开始为血库,糖尿病人群提供分析仪,公司不断扩大,期间也有收购其他公司并于2010年在英国上市。主要业务为床边检测设备,至今已生产超过8万台血红蛋白分析仪、糖化血红蛋白分析仪等在全球100多个国家使用。以其易用、可靠、精确的品牌形象赢得广泛赞誉。
ulian Baines在2000年进行了管理层收购,2008年使公司营业额达到8500万英镑。2009年12月Julian Baines正式成为集团的首席执行官,随后在2010、2011年和2014年成功完成三轮融资,在七个国家收购并集成了八个企业。创造了从零到4000万英镑的收益。2017年1月27日,因其在生命科学领域做出的贡献被授予英帝国员佐勋章。
EKF Diagnostics公司虽然成立时间不算太久远,但在POTC市场上也是一个发展迅速,相对历史悠久的公司。以易用、可靠、精确的品牌形象赢得了广泛赞誉,2010年后相继收购多家医药公司,资源优化整合形成了多样化,先进的一系列产品。涉及血红蛋白即时检测,糖尿病监控,妇女健康,运动健康,中心实验室,临床化学,生命科学等多个领域,其中血红蛋白分析仪EKF公司占据了最大的市场份额。此外EKF公司还有最权威的诊断专家,定时发布相关的诊断指南和标准。
公司产品
1、Diaspect Tm 血红蛋白分析仪
是EKF诊断公司旗下最新研发的一款便携式血红蛋白分析仪,也是现今世界上速度最快的血红蛋白分析仪。使用微玻片技术吸取样本,实现1秒极速检测。内置长寿命可充电电池,一次充电可使用40天,特殊的微玻片在开启包装后都可持续使用至有效期结束,不受湿度或温度的影响,因此尤其适合在炎热和潮湿的条件下使用。是一款胜任所有场合下的高精度快速血红蛋白分析仪系统。

2、BIOSEC_Line系列血糖乳酸分析仪
历经20余年的积累和完善,已成为该领的标志性产品。该系列葡萄糖乳酸分析仪目前有5样本、20样本和63样本三种型号可供用户选择。 Biosen分析仪有葡萄糖单通道和双通道系统,双通道系统还可以同时测量乳酸。Biosen使用特殊的传感器芯片实现快速测量保证了高精度性又降低了成本。由于传感器芯片系统几乎不需要维护,又具有触摸屏和大内存的特点极大的增加了Biosen分析仪的用户友好性。
(1)可使用全血、血浆、血清、尿液脑脊液等样本
(2) 25秒/Test 快速即时检测
(3)精度CV<1.5,偏差<3%

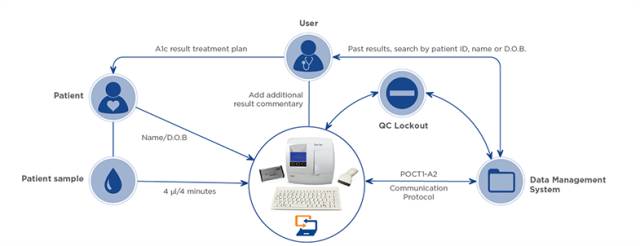
3、Quo-Test 糖化血红蛋白分析仪
是由EKF集团下属Quotient Diagnostics 公司研制。Quo-Test 糖化血红蛋白分析仪采用亲和荧光猝灭原理,检测过程十分简单,只需将样本用配套毛细管转移到试剂盒中即可上机操作。试剂盒采用一体化设计,无需处理样本和试剂,将人为操作误差降至最低。现已获得IFCC和NGSP认证。
作为一家全球性的在现场测试和中央实验室制造方面有着悠久历史的大企业,EKF Diagnostics公司已经形成了自己的品牌形象以及稳定的市场份额。正如MediWales论坛指出的EKF Diagnostics公司的优势在于跨国研究和跨国生产还有来自德国,爱尔兰,美国及英国的多个国家的经验丰富的分析师和工程师团队以及在医疗诊断领域最权威的专家。但是跨国也带来了新的问题在2016年EKF Diagnostics公司的年度报告中就有提到,希望通过集中制造业降低成本。随着现场检测的市场需求越来越大,更多的公司加入现场检测领域,例如瑞典HemoCue血红蛋白分析仪、挪威小旋风糖化血红蛋白仪、TRACE便携式葡萄糖、乳酸分析仪等,打江山容易,守江山难,期待EKF Diagnostics公司在未来的表现。
来源:火石创造
扫描下方二维码,关注Medtec China公众号,国际前沿医疗器械设计与制造深度好文一手掌握!












